
The Clinical Trial Design
BY DAN FRIEDMANN, MD
BOARD-CERTIFIED DERMATOLOGIST
This third-party, before-after clinical trial was designed to test the efficacy and safety of The Flasher™ 2.0, Nood's at-home, handheld intense pulsed light (IPL) device. Participants were followed in-clinic at Austin’s Westlake Dermatology over sixteen weeks.
Each participant performed 10-minute treatments at home on a single armpit twice weekly for twelve weeks. They visited Westlake Dermatology after four, eight, and twelve weeks of treatment to measure hair reduction and patient satisfaction and one final visit four weeks after their last treatment.
THE PARTICIPANTS
SAMPLE SIZE
40 People
AGES
18-45
TIMEFRAME
4 Months
92% Saw Hair Reduction
Hair reduction was measured by comparing hair regrowth between untreated vs treated underarms during in-clinic follow-up visits.
92% of participants rated hair reduction as improved, much improved, or very much improved after just 8 weeks of treatment.
LONG TERM HAIR REDUCTION
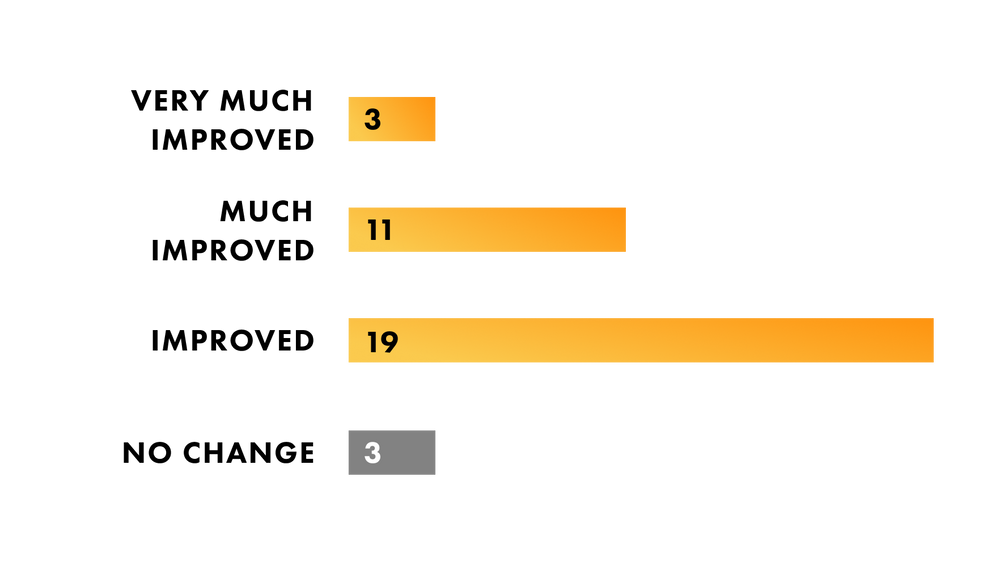
LONG TERM HAIR REDUCTION


Before & after images from twelve week third-party clinical trial. Both photos taken one week after shaving.
97% Were Satisfied
Overall treatment satisfaction was recorded during in-clinic follow-up vists at four, eight, and twelve weeks of treatment, plus four weeks after treatment.
97% of participants reported moderate, good, or very good satisfaction after just 8 weeks of treatment.
PARTICIPANT SATISFACTION

PARTICIPANT SATISFACTION


Before & after images from twelve week third-party clinical trial. Both photos taken one week after shaving.
Low Discomfort Levels
Users were asked about their average discomfort levels while using The Flasher™ 2.0 set to the highest level, seven, halfway through the 12-week treatment period. They rated their pain by marking a validated horizontal analog scale (with 0 being ‘no pain’ and 10 being ‘intolerable pain’).
The average discomfort level was < 1.


Visible Results
BEFORE AND AFTER IMAGES FROM OUR CLINICAL TRIAL


REBECCA K. | AGE: 24


KELLY J. | AGE: 32


MEGAN F. | AGE: 28






